A rapid visual categorisation task using natural images.
Images are displayed for a fraction of a second. Tap to classify each in turn as animal or non-animal.
A rapid visual categorisation task using natural images.
Images are displayed for a fraction of a second. Tap to classify each in turn as animal or non-animal.

Stimulus

Interval

Mask

Animal?

The Integrated Cognitive Assessment (ICA) test is based on humans’ strong reaction to animal stimuli, and the ability of a healthy brain to process images of animals in less than 200 ms.
The human brain is very sensitive to images of animals. There’s a good reason for this – ancient humans either had to run away from them or hunt them down for food. We call this the ‘food or fear’ factor. The test targets brain areas that are affected in the initial stages of cognitive disorders before severe memory symptoms become apparent.
Furthermore, the ‘spot the animal’ task makes ICA intrinsically independent of language and culture. Even non-human primates are able to perform animal/non-animal categorisation tasks like this one.
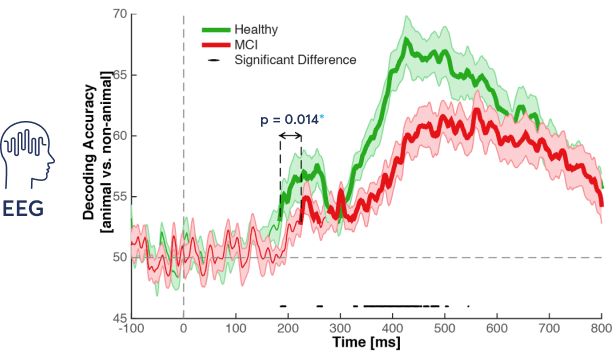
The EEG data from 100ms before to 800ms after the stimulus onset are extracted. A linear classifier is trained and tested at each time point, by cross-validation, to readout brain information available for decoding animacy.
*A p-value smaller than 0.05 is considered significant difference.
We recorded EEG data while people took the ICA test. Participants with mild cognitive impairment (MCI) showed a significant decrease in speed and accuracy of processing animacy information compared to healthy controls.
There is a clear difference in the level of activation between the three arms, giving us a neuroimaging basis for the discriminating power of ICA.
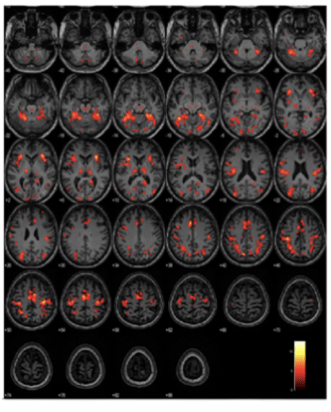
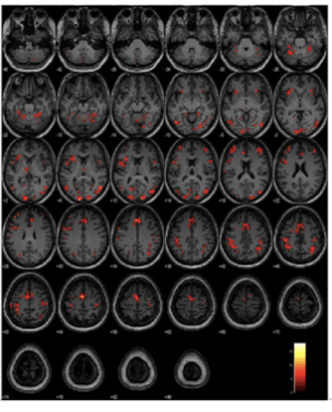

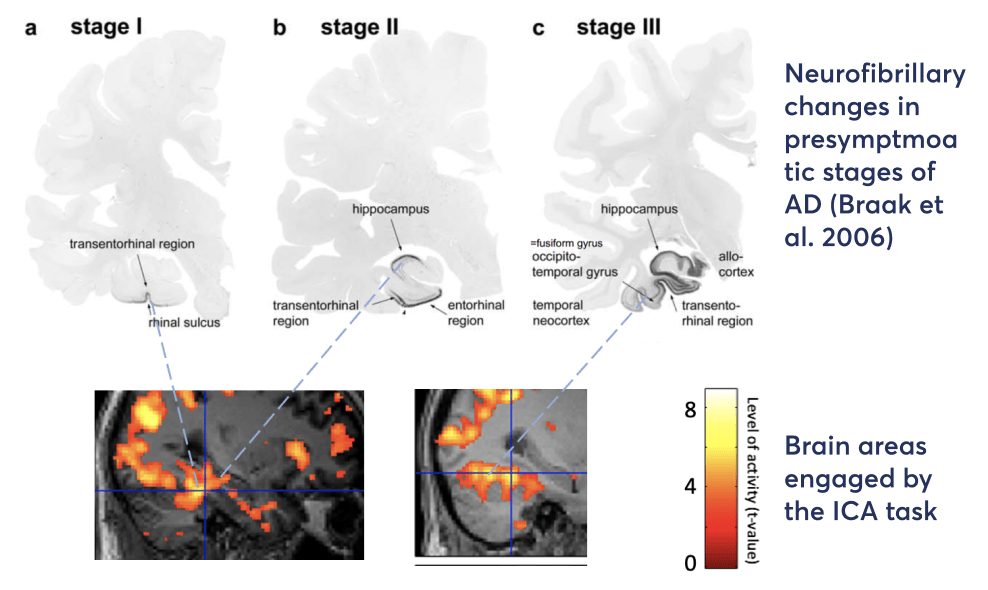
In a task-based fMRI study we find that the ICA task engages brain areas such as the transentorhinal region, fusiform gyrus and the inferior and middle temporal gyri, that are anatomically identified as being among the earliest areas affected in pre-symptomatic stages of AD, as shown by Braak and colleagues 1991 and 2006.
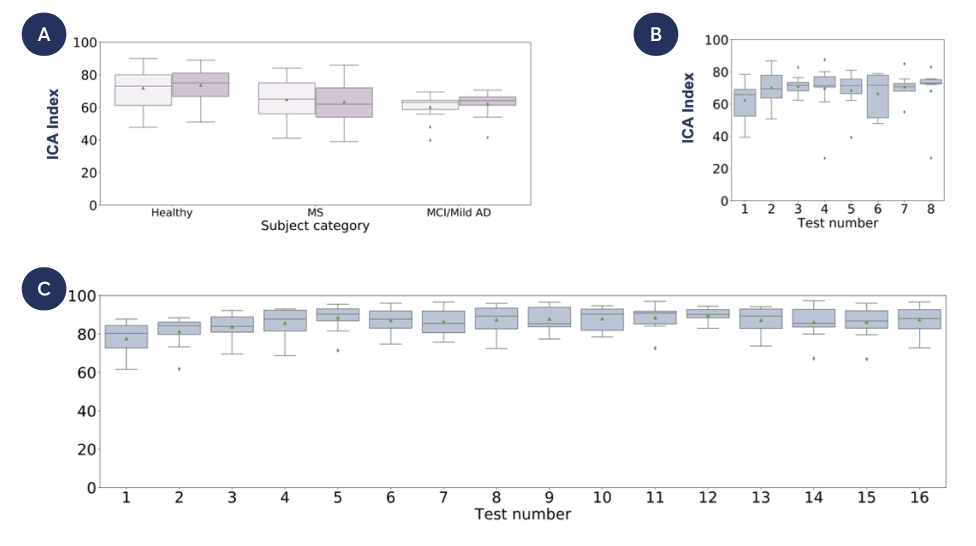
A. Test-retest scores for categories of participants: healthy (paired t-test p=0.06), MS (paired t-test p=0.18) and MCI/Mild AD (paired t-test p=0.43)
B. Healthy participants took the ICA test every other day for over two weeks. ANOVA one-way p-value=0.7
C. Healthy participants completed 16 tests on iPad and iPhone devices. ANOVA one way p-value=0.06
The ICA can be used repeatedly by the same individual, with no learning effect. This makes cognitive monitoring more accurate than standard tests.
The ICA can generalise across populations without the need for collection of population-specific normative data.
SM Khaligh-Razavi et al, Scientific Reports, volume 9, Article number: 1102 (2019).
This was demonstrated by the ability of the ICA to detect patterns of cognitive impairment that are common across cohorts of different cultural and demographic characteristics. Conventional pen and paper and computerised tests require re-norming and validation in different languages in order to be validated, requiring collection of culture-specific normative data before a test can be used in populations with different demographic characteristics. Both are prerequisites for large population deployment and risk-based screening in primary care.
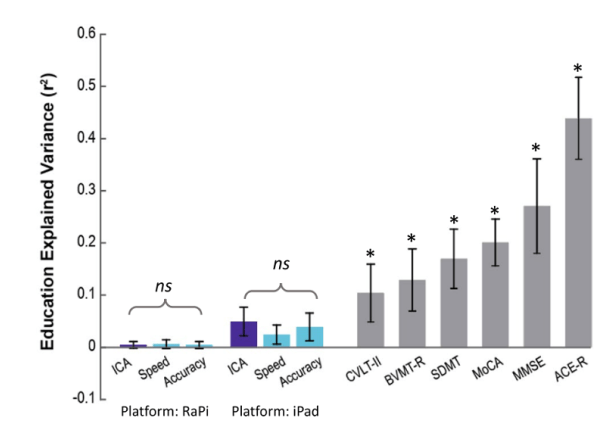
People with higher levels of education tend to score better in the standard pen-and-paper tests, compared to their age-matched group that fall into the same cognitive category. This makes ‘the level of education’ a confounding factor for cognitive assessment.
Thanks to our AI engine, the ICA is always improving in sensitivity and scope. This gives clinicians confidence in the model, protect against bias and can be used to improve the performance of the system over time.
The use of AI in decision making, particularly for diagnostic decisions in healthcare, requires a level of explainability from the model which can be used to understand the important factors which led to its output. This is in contrast to ‘black box’ models that offer limited interpretability of results and therefore prohibit their use in clinical practise.
Existing diagnostic tools remain static and never change. Through machine learning, our AI engine can be continually updated. Plus, the AI can be trained to test for other neurodegenerative conditions.
Our AI-powered system can take various patient attributes into account, including sex, age, education level through EHR integration. Current tools lack the capacity to incorporate the wealth of relevant data that is crucial to generate meaningful and accurate results for diagnosis.
The earlier you start looking after your cognitive health, the greater impact you’ll have.
Trouble remembering, concentrating, learning new things or making decisions.